Imaging Neuroinflammation in Neurodegeneration
Project Overview
Neuroinflammation is a fundamental biological process driving the progression of major neurodegenerative disorders, including Alzheimer’s disease, frontotemporal dementia, Parkinson’s disease, and ALS. Using advanced PET neuroimaging, our Neuroimaging Core investigates how microglial activation, astroglial responses, and inflammatory cascades contribute to neuronal injury and clinical decline. A comprehensive review of PET neuroinflammation literature highlights the essential role of PET imaging in mapping inflammatory changes in vivo and underscores the rapid expansion of inflammation‑targeted imaging research across diseases.
Scientific Focus Areas
1. High‑Resolution PET Imaging of Microglial Activation
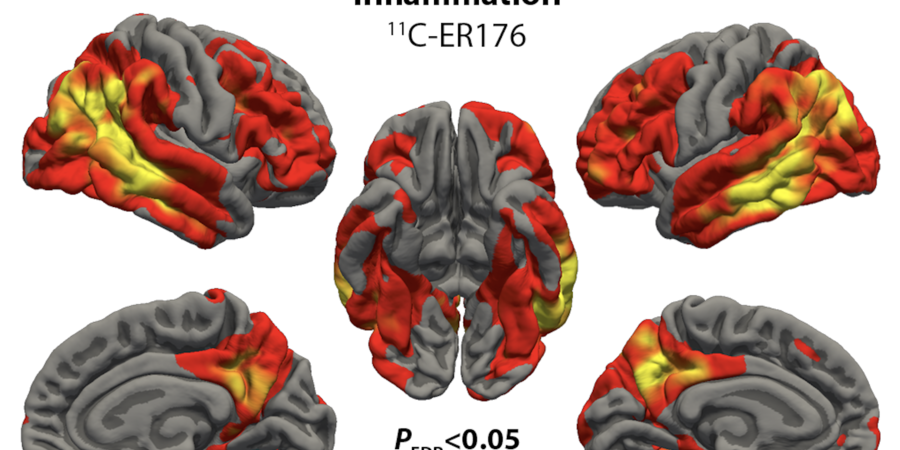
The Neuroimaging Core employs state‑of‑the‑art TSPO PET tracers, including 11C‑PBR28 and the next‑generation 11C‑ER176, to quantify neuroinflammation with high specificity. TSPO PET is the most widely used method for measuring neuroinflammation in vivo. The novel tracer 11C‑ER176 enables reliable imaging across all TSPO genotypes and reduces radiation exposure because of the short half‑life of Carbon‑11.
These tools allow us to map inflammation at unparalleled spatial resolution and relate it to structural, metabolic, and clinical biomarkers.
2. Inflammation Mapping in Frontotemporal Degeneration
Our work in semantic variant primary progressive aphasia demonstrates that neuroinflammation concentrates in regions undergoing active degeneration, including the temporal pole, insula, and orbitofrontal cortex. Distinct spatial patterns between inflammation (11C‑PBR28) and tau pathology (18F‑flortaucipir) indicate that inflammation may influence disease spread rather than simply follow it.
This research offers critical insight into how inflammatory processes shape frontotemporal lobar degeneration trajectories.
3. PET Biomarkers in Immunomodulation Clinical Trials
The Core supports biomarker‑driven clinical trials targeting inflammation, including IL‑2 immunotherapy in Alzheimer’s disease. Although TSPO‑PET showed stable inflammation before and after treatment, IL‑2 significantly increased T‑regulatory cell activity and improved CSF biomarkers such as Aβ42. This indicates that therapeutic immune modulation may produce biological improvements not immediately reflected in PET inflammation signal.
These results emphasize the need for multimodal biomarkers to fully capture therapeutic effects.
4. Foundational Neuroimaging Contributions
Neuroimaging Core investigators have made foundational contributions, including studies mapping large‑scale brain networks of the temporal pole and elucidating neuroimaging‑based differentiation of Alzheimer’s disease from vascular dementia. These findings provide the structural and functional framework supporting current inflammation‑focused research.
Impact
Through advanced PET imaging, quantitative modeling, and integration with CSF and blood biomarkers, the Neuroimaging Core advances understanding of how inflammation emerges and spreads in neurodegeneration, how it interacts with proteinopathies such as tau and TDP‑43, and how therapeutic interventions modulate immune activity in the brain. Our work establishes inflammation not only as a biomarker of disease activity but also as a promising therapeutic target, supported by emerging tracer technologies and high‑resolution imaging platforms.